Presentation
This session completes the high-temperature solid oxide fuel cell SOFC model presented in the sessions S61En and S62En.
The models we have developed in these sessions only allowed for pure hydrogen as fuel.
In reality, it is very rare that hydrogen is available: it must be produced from another fuel. This is called reforming.
Reforming a fuel such as methane can be modeled by the basic equation:
CH4 + H2O→ CO + 3 H2(ΔH = 206 140 kJ/kmol)
It allows one to converts the fuel into hydrogen, but requires water and heat supply, and produces carbon monoxide, which is a poison for some fuel cells such as PEMFC, where it is necessary also to convert CO.
These questions are the subject of the session S64En. Here, we will limit ourselves to a simplified approach to reforming, valid for high temperature and CO-insensitive SOFC or MCFC.
We will present two models of SOFC consuming methane, as well as an exercise of advanced modeling concerning a system developed by Siemens Westinghouse.
(Session realized on 09/14/11 by Renaud Gicquel)Fourth model
We will begin by refining the model developed in sessions S61En and S62En, considering that hydrogen is replaced by a mixture of methane and water in sufficient quantity to ensure complete conversion of methane.
We will introduce some improvements to the previous model:
- The electrical parameters of the equation, the fuel use rate and the cooling load will be editable
- A test will be conducted to check that there is enough oxygen in the combustion air to ensure the smooth running of the fuel cell
This model is presented in eight steps, other explanations are presented in the sessions S61En and S62En:
- The first three steps establish the chemical reactions that take place at the cell anode and cathode and determine the energies put into play;
- The next three steps show the results displayed on the stack component screens;
- The seventh step gives the composition of gases at the stack inlet and outlet, for the settings chosen;
- The last step presents the changes induced in the computer implementation of the externel class.
In Thermoptim, the classes representing the stack are SOFCCH4inlet and SOFCCH4outlet.
Explanations about this model are presented in Part V of the book Energy Systems, and all the work files for this example can be downloaded from the links below.
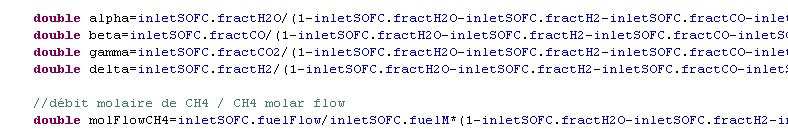
Anode side reactions
- methane reforming
- τ rate of fuel use
- part used:
- part non used:
- composition at the anode outlet:
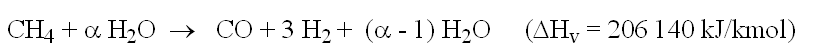
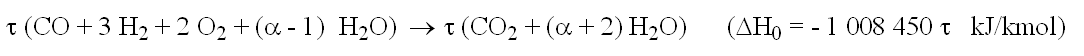
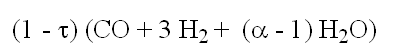
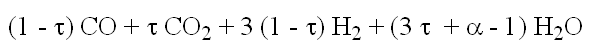
Cathode side reactions
- air is depleted by the oxygen used (2 τ)
- inlet air:
- outlet depleted air:
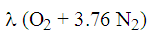
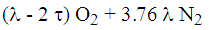
Energies put into play
- enthalpie released: τ ΔH0
- electricity: ε τ ΔH0
- heat for steam cracking: ΔHv
- thermocoupler ang gas heating: τ (1 - ε) ΔH0 - ΔHv
Synoptic view of the SOFC cell
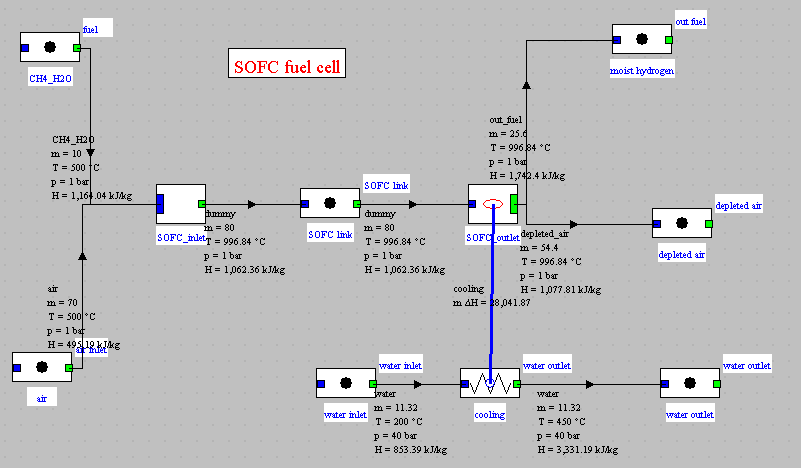
Upstream mixer screen
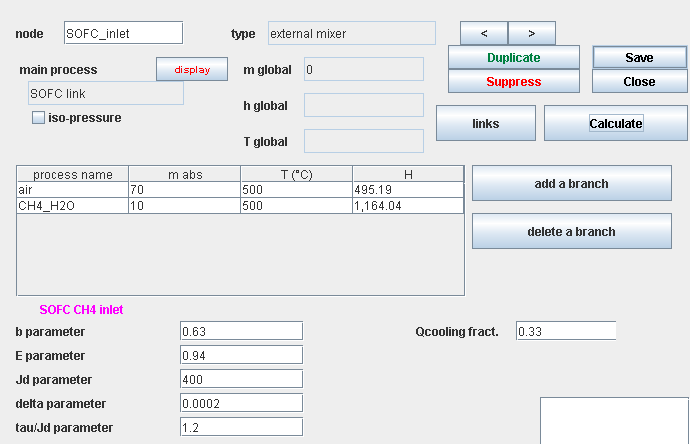
SOFC component screen
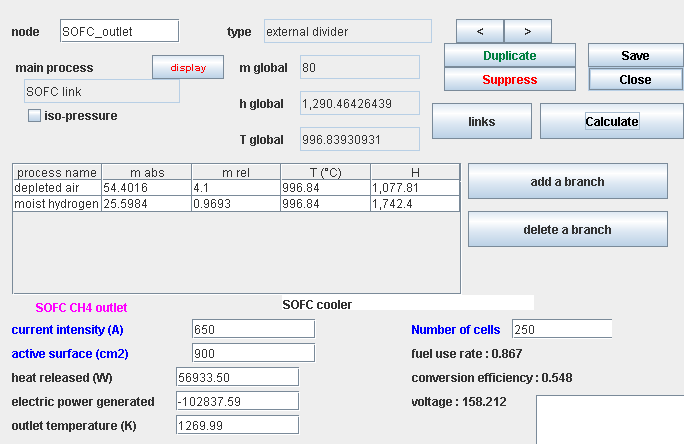
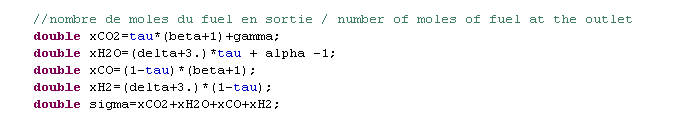
Composition of gases
- humidified inlet fuel, flow 10 g/s LHV: 22 562 kJ/kg (α = 1.083)
- humidified outlet fuel, flow 25.6 g/s LHV: 1 477 kJ/kg
- air depleted in O2, flow 64.4 g/s


Class code changes


Industrial module
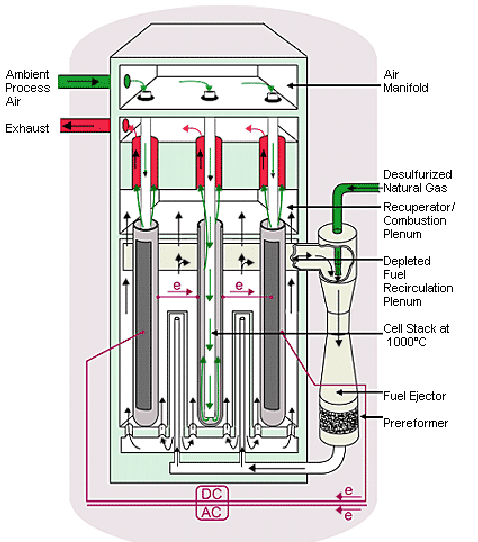
Influence of pressure
Doc. Siemens Westinghouse
Fifth model
We will now complete the model so that the water humidification can be provided by recycling a fraction of the gases leaving the anode.
This requires slightly modifying the structure of the inlet mixer, which must be able to identify the fuel branch thanks to the presence of methane. The mole fractions of the components present in the gases leaving the anode must be loaded into variables.
We should also take into account the influence of pressure on the performance of the stack.
The equations involved are given in the document Model5.pdf downloadable from one of the links below.
All the working files for this example can be downloaded from one of the links below.
Diagram with gas recycling
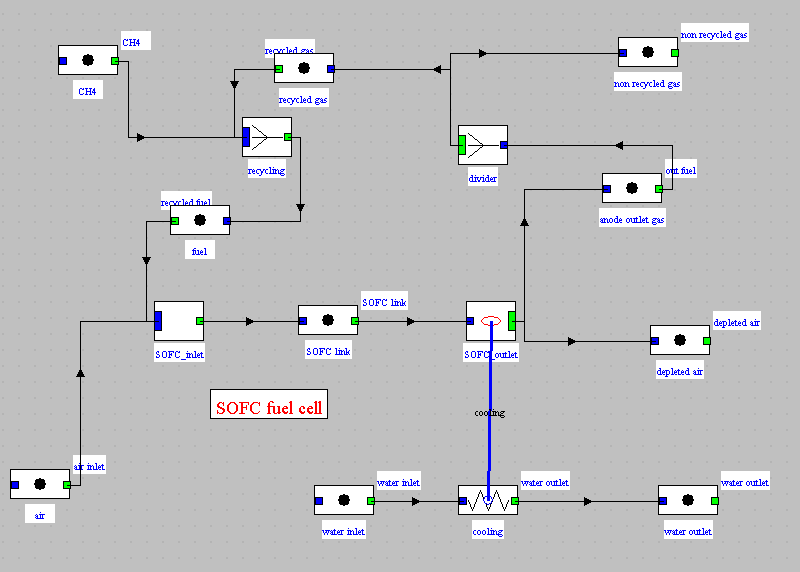
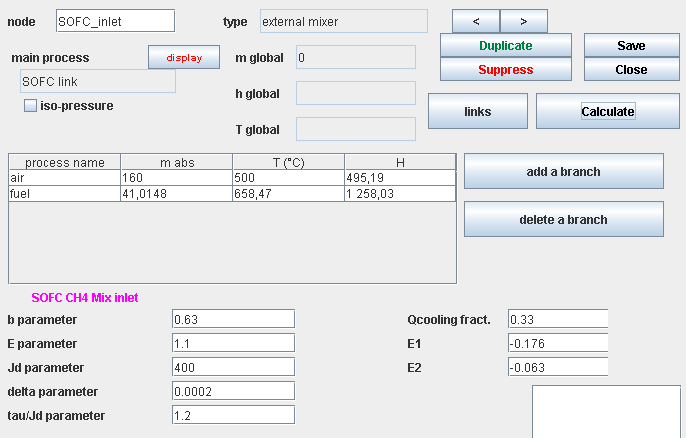
Upstream mixer screen
- influence of P : E = E0 + E1 exp(E2 P)
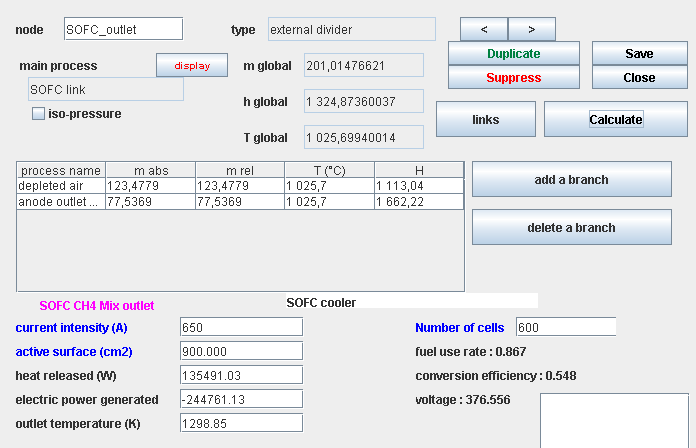
SOFC cell component screen
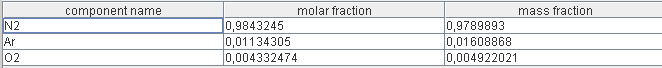
Composition of gases
- humidified inlet fuel, flow 41 g/s LHV: 13 056 kJ/kg
- humidified outlet fuel, flow 77.5 g/s LHV : 1 142 kJ/kg
- air depleted in O2, flow 123.5 g/s


Class code changes
- calculation of compositions
- Calculation of the open circuit voltage
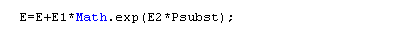
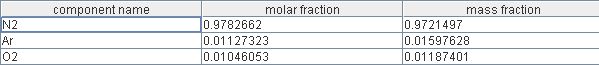
Modeling of the Siemens Westinghouse cell
In this exercise, whose solution is not provided, we suggest you start from the fifth model to model as closely as possible the system proposed by Siemens and Westinghouse and presented in one of the previous steps.
You will start by determining a set of parameters for the electrical model representing as well as possible the performance of cells whose polarization curves were given in this session, taking into account the influence of pressure.
Then you will represent all of the heat exchange taking place in the Siemens Westinghouse SOFC module, by adjusting the flows of methane, air and recycled gas to get the best performance. The final oxidation will be represented by a combustion chamber producing complete combustion of the gases leaving the anode not recycled.
According to the explanations available, you will determine whether or not to take into account the cooling of the stack, knowing that a setting in the inlet mixer screen allows you to do this.
The setting out of the exercise can be downloaded from the link below
Summary
In this session, we first develop a model of methane-fueled SOFC previously moistened, and we presented the results.
We then gave some details of the technological solutions adopted by Siemens Westinghouse, which has led us to further refine our model by adopting a more realistic approach, where water humidification is provided by recycling a fraction of anode outlet gases.
To consider the influence of pressure on the performance of the stack, we slightly modified the electric model presented in the session S62En.
We finally proposed an exercise (even a mini-project) which aims to finely model the Siemens Westinghouse system introduced.